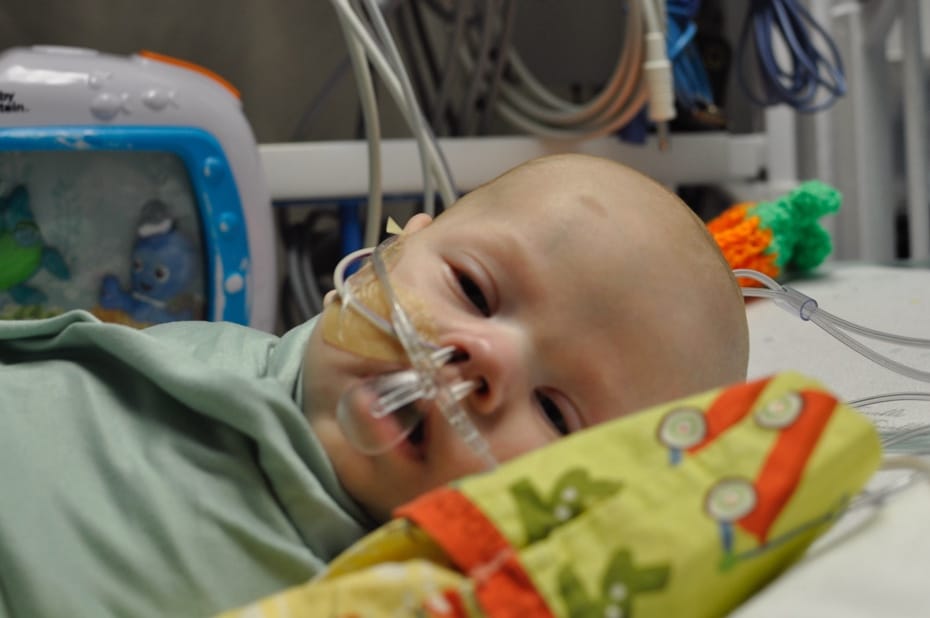
A bit over a month ago, I was set to testify before the House and Senate Safe Food Caucus in D.C.. Instead, the night before I had a heart attack. I am fine. I got great care at Howard University Hospital and have made a complete recovery – although changing a few habits going forward.
I had been asked to share some thoughts on the infant formula industry’s inability to make sure that such a critical food item is safe. As I sit here on this Sunday afternoon I am puzzled how we can spend countless billions on yet another unnecessary war and even more billions on AI and robots to make most of us irrelevant and we cannot make infant food safer? Here is some of what I wanted to discuss. I am sure there are other ideas – I would love to hear them.
Infant formula has until recently been considered low risk for C. botulinum (and, then there are other pathogens – Cronobacter sakazakii, Salmonella and recently, Bacillus cereus of concern).
The recent C. botulinum outbreak and the mixed picture you get from looking at the multitude of WGS sequences on NCBI indicates that infant formula might be riskier. We need a new risk assessment of C. botulinum (and other pathogens) in infant formula and its constituent ingredients and how to control the pathogens in powdered infant formula (PIF). For C. botulinum we need more information about:
How often is infant formula and its constituents contaminated with C. botulinum spores?
Do we detect the pathogen when we need to? We need to assess if the current methods are good enough (sensitive enough)?
How often is a product contaminated with more than one strain of C. botulinum (multiple WGS profiles)? How many isolates (colonies on a plate) from each sample should be sequenced to detect all contaminants?
Should we routinely culture infant formula from cases of infant botulism when the child has been fed such a product? (I highly suspect that infant formula is causing many more cases of infant botulism than we know of at the moment).
How do we best control C. botulinum spores in infant formula. We need studies on methods to control C. botulinummore efficiently in infant formula.
Do we need new regulations and redefined best practices for the production of infant formula and detecting and culturing the organism from product, environment and patients?
Perhaps as a start:
Fund a risk assessment on the prevalence of C. botulinum spores, Cronobacter sakazakii, Salmonella and Bacillus cereus and sulfite reducing clostridia (as indicators) in:
a. raw milk
b. pasteurized milk
c. dairy powders – including whole milk powder, whey powder, whey protein concentrates, whey protein hydrolysates, nonfat dry milk, skim milk powder
A specific risk assessment on the prevalence of C. botulinum spores and sulfite reducing clostridia in Powdered infant formula
Risk assessment on the prevalence of C. botulinum spores in minimally processed (non-retort thermally processed) commercial baby foods fed to children <1 year of age
Risk assessment on the prevalence of C. botulinum spores in low moisture infant first foods (puffs, cereals (rice and oatmeal), peanut butter, etc.)
Processing strategies to minimize spores in PIF – investigate technologies to reduce spores in infant foods while maintaining nutritional standards
Impact of farming practices on the incidence of C. botulinum spores in milk
a. Prevalence of C. botulinum spores in silage, bedding, etc., at the farm
b. Milk collection on organic vs. conventional dairy farms
Improved resources for the tracking and following up of Infant Botulism cases to determine the source of botulism outbreaks
Bottomline for control of spores in powdered infant formula (PIF) either they destroy all the spores or reformulate to lower the incidence of spores in PIF (at least down to where it was prior to the addition of whole milk powder into the formulations of PIF). Because PIF is fed to infants from Day 1 it should be commercially sterile. Leave it up to the companies to determine how to make PIF commercially sterile. At the very least I would suggest that until a study is conducted to show the prevalence of C. botulinum spores in whole milk powder and a risk assessment is conducted, any formulation that contains whole milk powder should not enter into commerce.